Vascular Signal Transduction Mechanisms
There are several signal transduction mechanisms that modulate intracellular calcium concentration and therefore the state of vascular tone. Two important mechanisms will be described here: 1) G-protein-coupled pathway, and 2) the nitric oxide-cGMP pathway.
G-Protein Coupled Signal Transduction
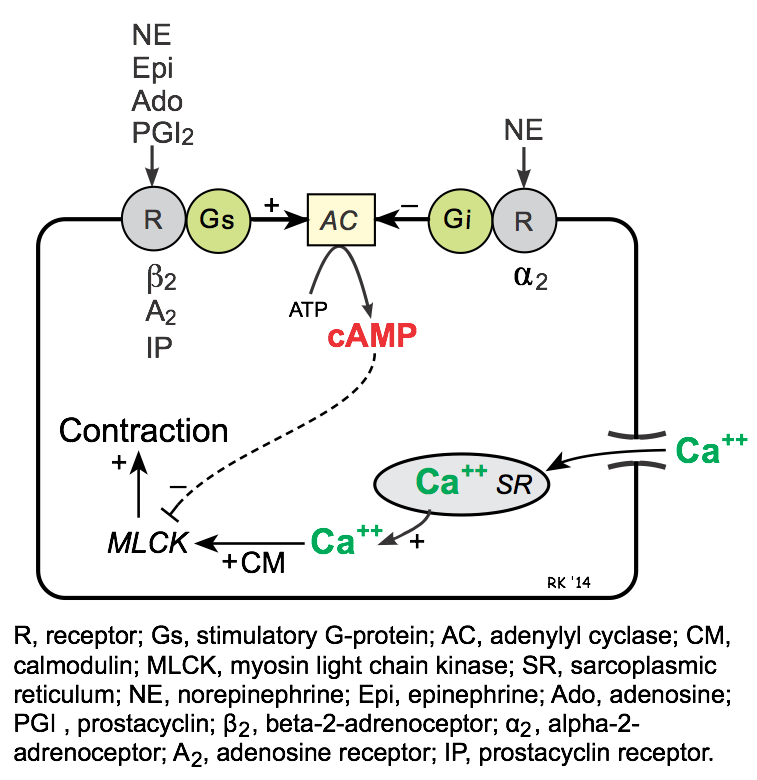 Like heart muscle, the Gs-protein coupled pathway in smooth muscle stimulates adenylyl cyclase (AC), which catalyzes the formation of cAMP. Unlike the heart, an increase in cAMP in vascular smooth muscle causes reduced contraction (i.e., relaxation). The reason for this opposite effect is that calcium-calmodulin activates myosin light chain kinase (MLCK) in vascular smooth muscle, which phosphorylates myosin and causes contraction; however, MLCK is inhibited by cAMP. In contrast to cardiac cells, Gs-protein stimulated increases in cAMP do not increase intracellular calcium.
Like heart muscle, the Gs-protein coupled pathway in smooth muscle stimulates adenylyl cyclase (AC), which catalyzes the formation of cAMP. Unlike the heart, an increase in cAMP in vascular smooth muscle causes reduced contraction (i.e., relaxation). The reason for this opposite effect is that calcium-calmodulin activates myosin light chain kinase (MLCK) in vascular smooth muscle, which phosphorylates myosin and causes contraction; however, MLCK is inhibited by cAMP. In contrast to cardiac cells, Gs-protein stimulated increases in cAMP do not increase intracellular calcium.
The Gs-protein is coupled to several important receptors that bind vasodilator substances, among which are β2-adrenoceptors (bind to β2-agonists such as epinephrine and isoproterenol), A2 purinergic receptors (bind to adenosine), and IP receptors (bind prostacyclin, PGI2).
Gi-proteins in vascular smooth muscle are coupled to α2-adrenoceptors. Binding of these receptors to an agonist such as norepinephrine reduces cAMP, which leads to smooth muscle contraction.
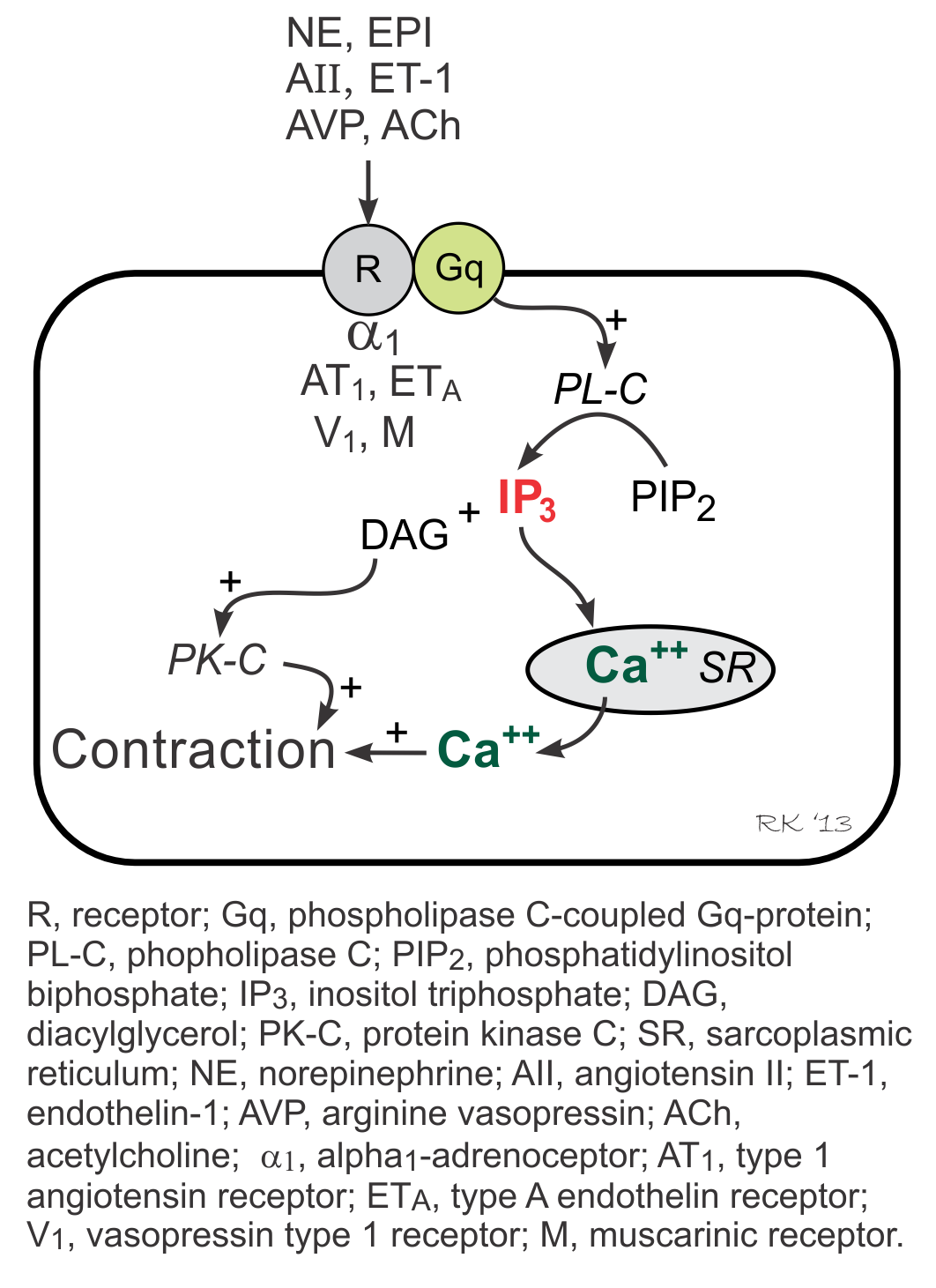 Gq-proteins in vascular smooth muscle are coupled to α1-adrenoceptors (bind to norepinephrine), ETA receptors (bind to endothelin-1), AT1 receptors (bind to angiotensin II), V1 receptors (bind to vasopressin), and muscarinic (M3) receptors (bind to acetylcholine). Two signal transduction pathways are linked to Gq-proteins: phospholipase C pathway (forms inositol triphosphate, IP3) and the Rho-kinase pathway. IP3 pathway stimulates SR release of calcium and activates protein kinase C (PK-C) via formation of diacylglycerol (DAG), which stimulates contraction. The Rho-kinase pathway (not shown in figure) inhibits myosin light chain phosphatase, which enhances contraction.
Gq-proteins in vascular smooth muscle are coupled to α1-adrenoceptors (bind to norepinephrine), ETA receptors (bind to endothelin-1), AT1 receptors (bind to angiotensin II), V1 receptors (bind to vasopressin), and muscarinic (M3) receptors (bind to acetylcholine). Two signal transduction pathways are linked to Gq-proteins: phospholipase C pathway (forms inositol triphosphate, IP3) and the Rho-kinase pathway. IP3 pathway stimulates SR release of calcium and activates protein kinase C (PK-C) via formation of diacylglycerol (DAG), which stimulates contraction. The Rho-kinase pathway (not shown in figure) inhibits myosin light chain phosphatase, which enhances contraction.
To summarize, the following agonists act directly on vascular smooth muscle through binding to their receptors, causing contraction via Gq-protein: norepinephrine, epinephrine, angiotensin II, endothelin-1, arginine vasopressin, and acetylcholine.
A special note regarding epinephrine. Its binding to Gs-protein-coupled β2-adrenoceptors versus Gq-protein-coupled α1-adrenoceptors is concentration dependent because β2-adrenoceptors have a higher affinity for epinephrine than α1-adrenoceptors. Therefore, at low epinephrine concentrations, vasodilation occurs, whereas at high concentrations, vasoconstrictor responses become dominant.
cGMP-Coupled Signal Transduction
A third mechanism that is important in regulating vascular smooth muscle tone is the nitric oxide (NO)-cGMP system. Vascular endothelial cells normally produce NO, which diffuses from endothelial cells to adjacent smooth muscle cells where it activates guanylyl cyclase, leading to increased formation of cGMP and vasodilation. Increased cGMP relaxation of vascular smooth muscle involves activation of cGMP-dependent protein kinase, inhibition of calcium entry into the vascular smooth muscle, activation of K+ channels (causing hyperpolarization), and decreased IP3.
Acetylcholine (ACh), whether administered intravascularly or released by cholinergic autonomic (parasympathetic) nerves, binds to muscarinic receptors (M3) on the vascular endothelium, which stimulates the formation and release of NO as described above to produce vasodilation. Although ACh binds to M3 receptors on both endothelial and vascular smooth muscle cells, if the endothelium is normal, then the formation of endothelial-derived NO and vasodilation overrides smooth muscle M3-mediated contraction. If the endothelium is damaged so that NO release is diminished, then ACh-induced vasodilation is changed to ACh-induced vasoconstriction.
Certain antihypertensive and antianginal drugs are called nitrodilators because they release NO, and mimic the effect of endothelial produced NO to produce vasodilation.
Revised 12/13/2022
 Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021)
Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021) Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)
Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)