Arterial Baroreceptors
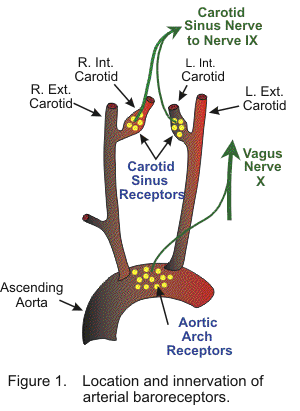 Arterial blood pressure is normally regulated within a narrow range, with a mean arterial pressure typically ranging from 85 to 100 mmHg in adults. It is important to control arterial pressure to ensure adequate blood flow to organs throughout the body. This is accomplished by negative feedback systems incorporating pressure sensors (i.e., baroreceptors) that sense the arterial pressure. The most important arterial baroreceptors are in the carotid sinus (at the bifurcation of external and internal carotids) and in the aortic arch (Figure 1). These receptors respond to stretching of the arterial wall so that if arterial pressure suddenly rises, the walls of these vessels passively expand, which increases the firing frequency of action potentials generated by the receptors. If arterial blood pressure suddenly falls, decreased stretch of the arterial walls leads to a decrease in receptor firing.
Arterial blood pressure is normally regulated within a narrow range, with a mean arterial pressure typically ranging from 85 to 100 mmHg in adults. It is important to control arterial pressure to ensure adequate blood flow to organs throughout the body. This is accomplished by negative feedback systems incorporating pressure sensors (i.e., baroreceptors) that sense the arterial pressure. The most important arterial baroreceptors are in the carotid sinus (at the bifurcation of external and internal carotids) and in the aortic arch (Figure 1). These receptors respond to stretching of the arterial wall so that if arterial pressure suddenly rises, the walls of these vessels passively expand, which increases the firing frequency of action potentials generated by the receptors. If arterial blood pressure suddenly falls, decreased stretch of the arterial walls leads to a decrease in receptor firing.
The carotid sinus baroreceptors are innervated by the sinus nerve of Hering, which is a branch of the glossopharyngeal nerve (IX cranial nerve). The glossopharyngeal nerve synapses in the nucleus tractus solitarius (NTS) in the brainstem's medulla. The aortic arch baroreceptors are innervated by the aortic nerve, which then combines with the vagus nerve (cranial nerve X) traveling to the NTS. The NTS modulates the activity of sympathetic and parasympathetic (vagal) neurons in the medulla, which regulate the autonomic control of the heart and blood vessels.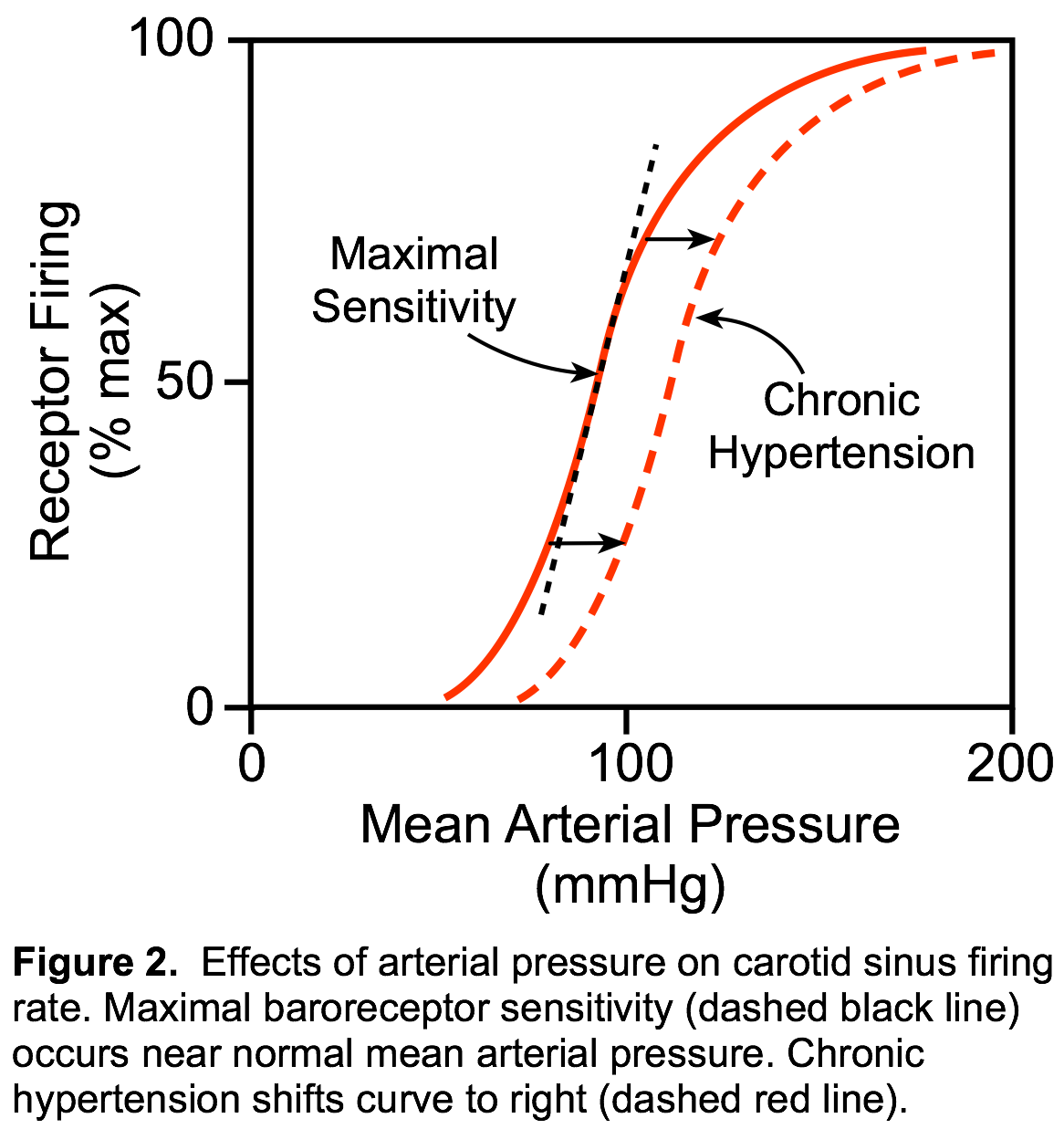 Of these two sites for arterial baroreceptors, the carotid sinus is quantitatively the most important for regulating arterial pressure. The carotid sinus receptors respond to pressures ranging from 60 to 180 mmHg (Figure 2). Receptors within the aortic arch have a higher threshold pressure and are less sensitive than the carotid sinus receptors. Maximal carotid sinus sensitivity occurs near the normal mean arterial pressure; therefore, very small changes in arterial pressure around this "set point" dramatically alter receptor firing so that autonomic control can be altered in such a way that the arterial pressure remains very near to the set point. This set point changes during exercise, hypertension, and heart failure. In chronic hypertension, the response curve shifts to the right, which increases the set point pressure. This explains, in part, how arterial pressure can remain elevated during chronic hypertension.
Of these two sites for arterial baroreceptors, the carotid sinus is quantitatively the most important for regulating arterial pressure. The carotid sinus receptors respond to pressures ranging from 60 to 180 mmHg (Figure 2). Receptors within the aortic arch have a higher threshold pressure and are less sensitive than the carotid sinus receptors. Maximal carotid sinus sensitivity occurs near the normal mean arterial pressure; therefore, very small changes in arterial pressure around this "set point" dramatically alter receptor firing so that autonomic control can be altered in such a way that the arterial pressure remains very near to the set point. This set point changes during exercise, hypertension, and heart failure. In chronic hypertension, the response curve shifts to the right, which increases the set point pressure. This explains, in part, how arterial pressure can remain elevated during chronic hypertension.
Baroreceptors are sensitive to the rate of pressure change besides the steady or mean pressure. Therefore, at a given mean arterial pressure, decreasing the pulse pressure (systolic minus diastolic pressure) decreases the baroreceptor firing rate. This is important during conditions such as hemorrhagic shock in which pulse pressure as well as mean pressure decreases. The combination of reduced mean pressure and reduced pulse pressure amplifies the baroreceptor response.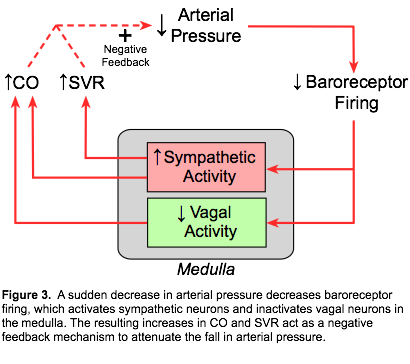 Although the baroreceptors can respond to either an increase or decrease in systemic arterial pressure, their most important role is responding to sudden reductions in arterial pressure (Figure 3). This can occur, for example, when a person suddenly stands up or following blood loss (hemorrhage). A decrease in arterial pressure (mean, pulse, or both) results in decreased baroreceptor firing. Autonomic neurons within the medulla respond by increasing sympathetic outflow and decreasing parasympathetic (vagal) outflow. Under normal physiological conditions, baroreceptor firing exerts a tonic inhibitory influence on sympathetic outflow from the medulla. Therefore, acute hypotension results in a disinhibition of sympathetic activity within the medulla, so that sympathetic activity originating within the medulla increases. These autonomic changes cause vasoconstriction (increased systemic vascular resistance, SVR), tachycardia, and positive inotropy (cardiac contractility). The latter two changes increase cardiac output. Increases in cardiac output and SVR lead to a partial restoration of arterial pressure.
Although the baroreceptors can respond to either an increase or decrease in systemic arterial pressure, their most important role is responding to sudden reductions in arterial pressure (Figure 3). This can occur, for example, when a person suddenly stands up or following blood loss (hemorrhage). A decrease in arterial pressure (mean, pulse, or both) results in decreased baroreceptor firing. Autonomic neurons within the medulla respond by increasing sympathetic outflow and decreasing parasympathetic (vagal) outflow. Under normal physiological conditions, baroreceptor firing exerts a tonic inhibitory influence on sympathetic outflow from the medulla. Therefore, acute hypotension results in a disinhibition of sympathetic activity within the medulla, so that sympathetic activity originating within the medulla increases. These autonomic changes cause vasoconstriction (increased systemic vascular resistance, SVR), tachycardia, and positive inotropy (cardiac contractility). The latter two changes increase cardiac output. Increases in cardiac output and SVR lead to a partial restoration of arterial pressure.
It is important to note that baroreceptors adapt to sustained changes in arterial pressure. For example, if arterial pressure suddenly falls when a person stands, the baroreceptor firing rate will decrease; however, after a period of time, the firing returns to near normal levels as the receptors adapt to the lower pressure. Therefore, the long-term regulation of arterial pressure requires activation of other mechanisms (primarily hormonal and renal) to maintain normal blood pressure.
Revised 12/7/2022
 Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021)
Cardiovascular Physiology Concepts, 3rd edition textbook, Published by Wolters Kluwer (2021) Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)
Normal and Abnormal Blood Pressure, published by Richard E. Klabunde (2013)